Industry News
News & Articles
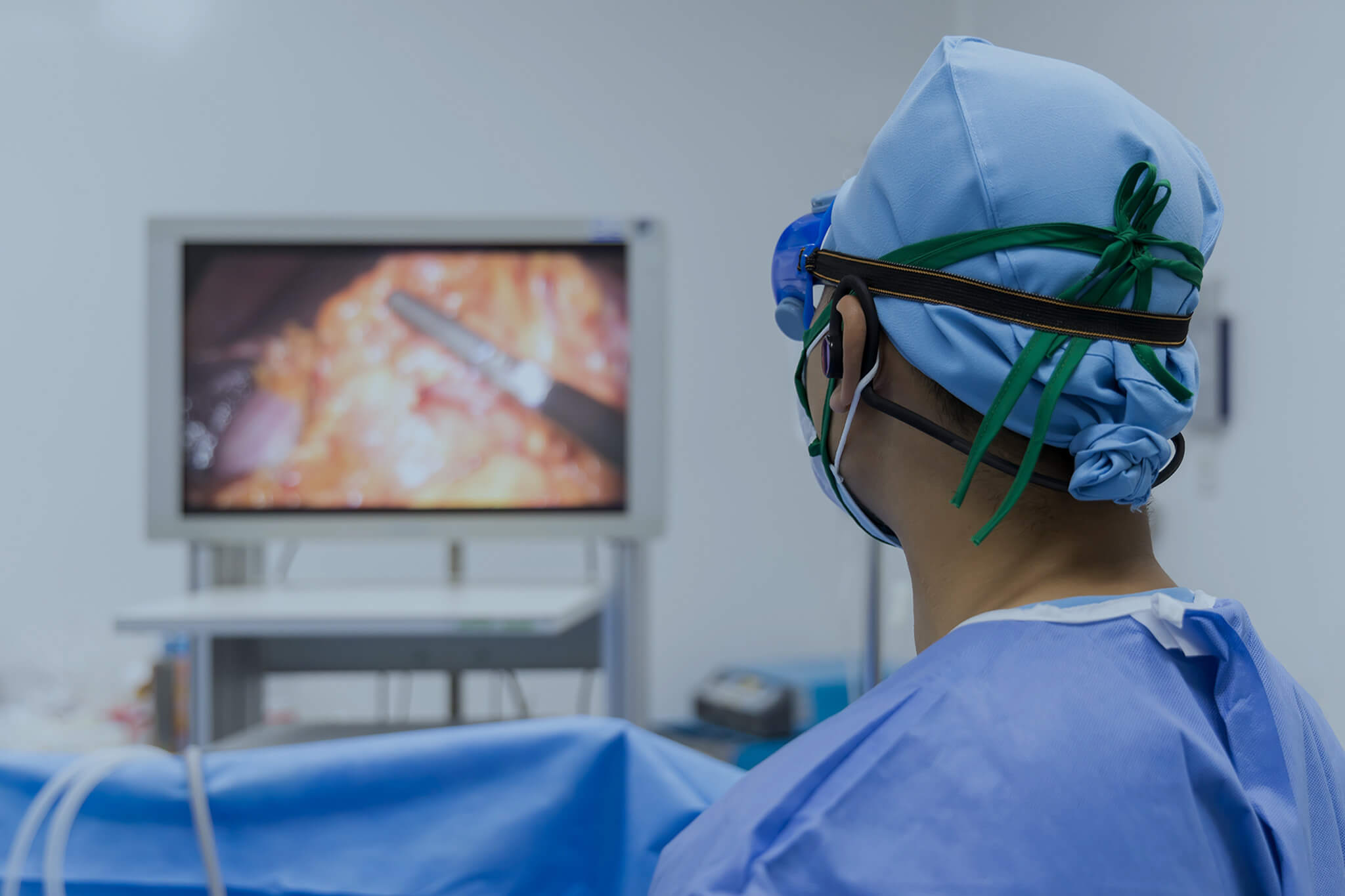
Endoscopic Injection Procedure
Learn how the endoscopic injection procedure is performed and what it treats, including the role of sterile injection needles and key safety considerations.

Mallory-Weiss Tear Treatment
Laser‑assisted Mallory‑Weiss tear treatment controls bleeding fast, lowers re‑intervention rates, and trims OR time.

GI Endoscopic Laser Therapy Procedure
Laser endoscopy lets GI teams stop bleeding, ablate tumors, and relieve strictures with pinpoint accuracy — faster, gentler care than many standard techniques.
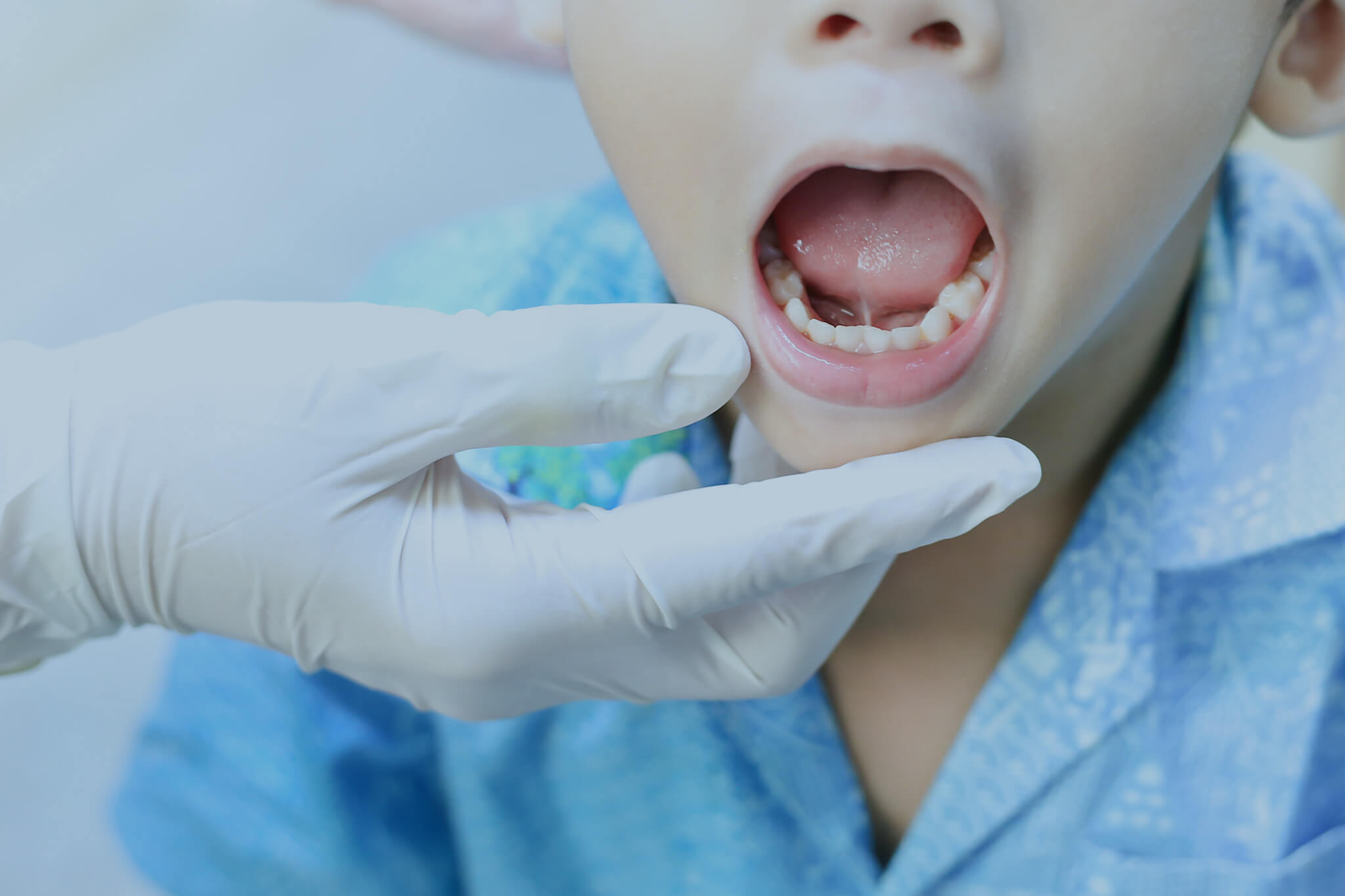
Laser Frenectomy Procedure
Discover how a frenectomy laser can streamline tongue-tie or lip-tie treatments, reduce healing time, and improve patient outcomes.

Laser Gingivectomy Procedure
Discover how laser gingivectomy removes and reshapes gum tissue for crown prep or aesthetics with less bleeding, minimal pain, and faster recovery in modern dentistry.

Laser Tonsil Cryptolysis Procedure
Discover how laser tonsil cryptolysis targets problematic crypts for tonsil stones and learn why CO₂ lasers are ideal for precise, minimally invasive cryptolysis.